 
For example, 2p (n+ l =2+1=3) is filled before 3s (n+ l =3+0=3) because 2p has lower value of n. (ii) For the orbital having the same value of n+l the orbital having glower value of n is filled up first. (i) Orbital are filled in order of increasing value of n+ l. Summary The Aufbau principle gives the order of electron filling in an atom. Since the energy of an orbital is determined by the quantum number n and l the sequence of filling the orbital proceeds according to the following rules: Therefore, according to Aufbau Principle :” In the ground state of an atom the electrons occupy the lowest energy orbital available to them,” This means that the electrons enter the orbital in the order of increasing energies he lowest energy available orbital being filled up first. The specific order of subshells using the Aufbau Principle can be found using the diagonal arrow diagram found in Chapter 8, and this is the same order found on the periodic. It is complicated by occasional overlapping. 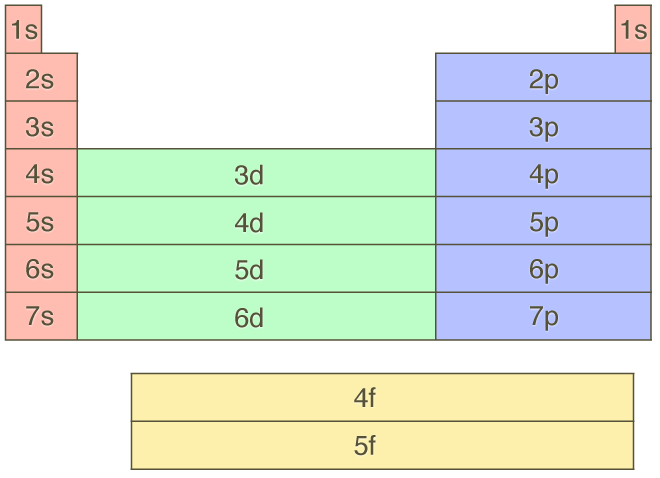
Higher numbered shells have higher energy. The assignment of electrons to orbital’s is done in order of their decreasing stability taking care not to violate the Pauli exclusion principle. The Aufbau Principle describes the order of subshells in an atom in terms of energy. A sample electron order filling diagram is shown in Figure 3 for calcium. Beyond the second energy level, the filling of atomic orbitals does not follow a simple pattern. The s-orbital is always the lowest energy sublevel. “building-up”) is the procedure applied to build up the ground state (minimum energy) electronic configuration by adding stepwise protons to the nucleus and electrons to the orbital stating with the hydrogen atom. The aufbau principle states that electrons enter orbitals of the lowest energy first. The Aufau principle (Aufbau is a German word meaning Arrangement of electrons in various orbital in an atom is called the electronic configuration of the atom. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
